'Interesting Timing': Fauci Touts Monoclonal Antibody Treatments One Day After FDA Approves Pfizer VaxChris MenahanInformationLiberation Aug. 27, 2021 |
Popular 
Netanyahu Working to Cement U.S. Aid to Israel Through 'Partnership'

DOJ Indicts Jewish Group for 'Large-Scale, Decade-Long Insider Trading Scheme'

Ben Shapiro: Trump Should 'Just Blow Up Kharg Island'

IDF Soldier Takes Sledgehammer to Jesus Statue During Operations in Lebanon

Mark Levin and Jonathan Pollard Push for Nuking Iran
  Pure coincidence, no doubt! Pure coincidence, no doubt!
From NY Post: COVID-19 sufferers can reduce their risk of hospitalization and death by up to 85 percent if they receive monoclonal antibody treatments in the early stages of their illness, Dr. Anthony Fauci said Tuesday.WATCH: Regeneron's treatment costs $1,250 a dose. The federal government is currently covering the costs. Several antibody treatments were greenlit for use in limited cases by the FDA through emergency use authorizations in May 2021 and Nov 2020. From the FDA in May: The data supporting this EUA for [GlaxoSmithKline's antibody treatment] sotrovimab are based on an interim analysis from a phase 1/2/3 randomized, double-blind, placebo-controlled clinical trial in 583 non-hospitalized adults with mild-to-moderate COVID-19 symptoms and a positive SARS-CoV-2 test result. Of these patients, 291 received sotrovimab and 292 received a placebo within five days of onset of COVID-19 symptoms. The primary endpoint was progression of COVID-19 (defined as hospitalization for greater than 24 hours for acute management of any illness or death from any cause) through day 29. Hospitalization or death occurred in 21 (7%) patients who received placebo compared to 3 (1%) patients treated with sotrovimab, an 85% reduction.The death rate in the placebo group of 7% strikes me as unusually high. It's interesting the way Big Pharma's drugs always seem to get Fauci's backing while cheap drugs studies indicate may be quite effective are blown off or derided. For example, Fauci touted the hell out of remdesivir which appears to have been a bust. The monoclonal antibody treatment appears to be only available for people who HHS considers "high risk."  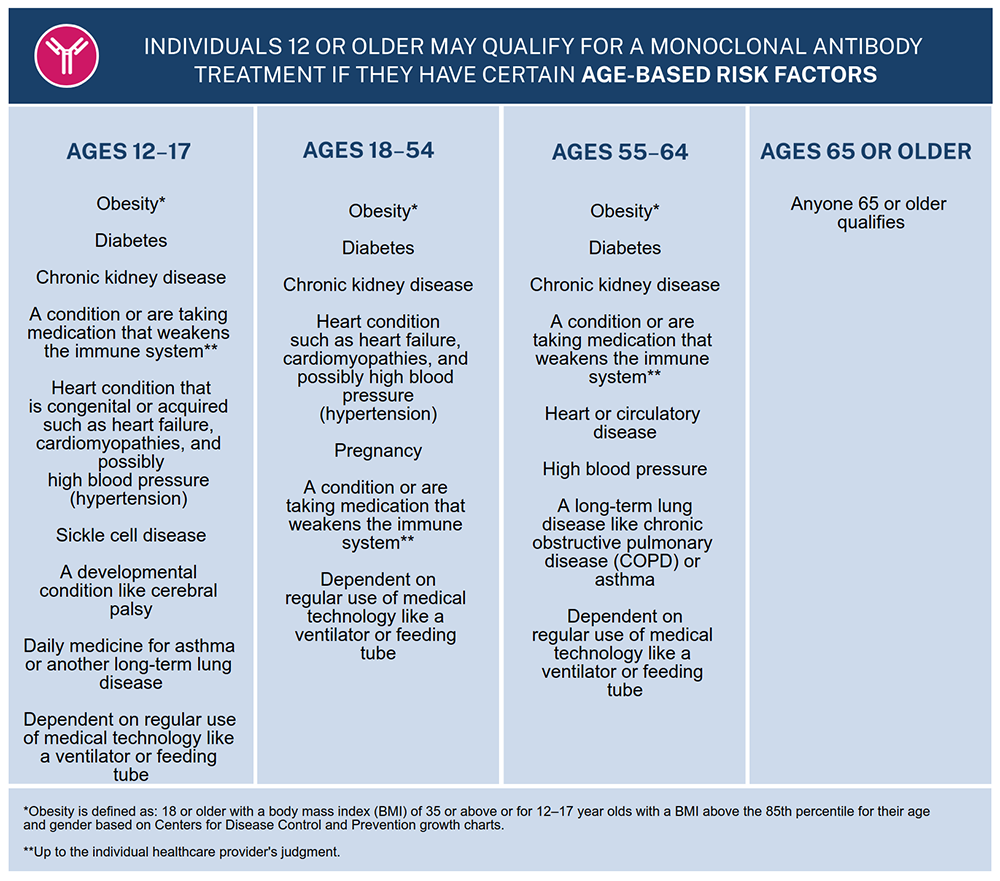 I can't help but wonder if the evidence piling up that vaccines appear to be failing is causing Fauci et al. to finally look to alternatives. Follow InformationLiberation on Twitter, Facebook, Gab, Minds, Parler and Telegram. |



