Unintended Consequences: New Formula of OxyContin Leads to Heroin Addiction and OverdoseStop OxyJan. 18, 2011 |
Popular 
Patriot ACT on Steroids: FISA Bill Forces 'An Enormous Range' of Businesses to Act as NSA Spies

Israel to Extend Water Agreement With Jordan 'For Helping Shoot Down Iranian Drones'

John Hagee Cheers Israel-Iran Battle as 'Gog and Magog War,' Will Lobby Congress Not to Deescalate

Report On HIAS-Linked NGO 'Encouraging Illegals to Vote Biden' Called Into Question
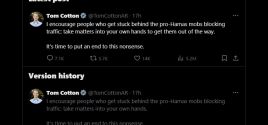
Sen. Tom Cotton Tells Followers to 'Take Matters Into Your Own Hands' Against 'Pro-Hamas Mobs'
 It has been almost a year since the U.S. Food and Drug Administration released their new formulation for OxyContin. The FDA released the new design of the tablet to help prevent the illegal misuse and abuse of the pain relieving drug. As successful as the new formula may be, the new version is causing authorities across the nation new stress. Authorities have been noticing a rise in more individuals turning to heroin to get the sensation of euphoria that at one time OxyContin was able to fulfill. On April 5, 2010 the U.S. Food and Drug Administration approved the new formulation of the time controlled-release drug OxyContin. OxyContin is made to slowly release potent opioid oxycodone into patients that require management of a continuous around-the-clock pain suppressant. However, because of OxyContin's time-released formula, each tablet contains large doses of oxycodone. With the old formula, individuals could release the high levels of oxycodone all at once. This leads to the illegal misuse and abuse of the tablet, which have resulted in thousands of fatal overdoses. The new formulation has been designed to prevent the tablet from being; cut, broken, crushed, or dissolved by individuals that are looking to misuse the drug. The U.S. Food and Drug Administration are aware that the new formulation of the tablet is not foolproof. Although the FDA feels that the new tablet is an improvement to help prevent future overdoses due to snorting, injecting or ingesting larger doses than recommended. Now that the tablet is harder (but not impossible) to tamper with,Ā drug users are looking for the other options to get the same affect. Read More |



